Clinicians and care teams
Explore how Regenemm Voice supports clinical documentation, patient summaries, handover quality and clinician-reviewed outputs.
Explore Regenemm VoiceThe AI-native parent company behind Regenemm Healthcare.
Founded in 2019, CTI builds clinically grounded AI systems for healthcare.
From our early work in stress-performance monitoring, body movement analysis, biosensors, sleep, and circadian health, CTI has evolved into the parent company behind Regenemm Healthcare: a hub-and-spoke clinical AI ecosystem with a secure, consent-aware, audit-grade control plane designed for visibility, transparency, and human review.
Creative Thinking Institute is the AI-native parent company behind Regenemm Healthcare. CTI began with the problem of human performance under stress, then expanded into physiology, sleep, circadian rhythm, wearable biosensors, soft sensing, rehabilitation movement analysis, and medical IoT data streams.
The GPT era accelerated the mission. CTI did not pivot from nothing into AI; it extended a pre-existing machine-learning, biosensor, and clinical-data thesis into AI-native healthcare infrastructure with human oversight, provenance, consent and public accountability as operating requirements.
CTI explains the research and governance layer. Regenemm shows how that work becomes clinical AI infrastructure in practice.
Explore how Regenemm Voice supports clinical documentation, patient summaries, handover quality and clinician-reviewed outputs.
Explore Regenemm VoiceFollow CTI's research lineage across stress-performance, biosensors, movement analysis, sleep, medical IoT and clinical AI.
View research writingStart with CTI's parent-company governance stance, then follow the product evidence through Regenemm's trust, security and clinical safety surfaces.
Start with CTI governanceRead current CTI essays on consent, shared transparency, interoperability, human oversight and evidence-seeking healthcare AI.
Read latest writingCTI is the origin, governance, research, and credibility layer. Regenemm Healthcare is the platform. Shared transparency is the product.
CTI's AI strategy started before the chatbot era: with structured health data, wearable signals, psychometrics, machine learning, consent, and clinical context.
The Regenemm Hub governs identity, consent, audit evidence, exports, persistence, cross-spoke routing, entitlements, telemetry, and compliance evidence.
AI lifecycle controls, impact assessments, system cards, data lineage, release gates, monitoring evidence, and executive risk ownership are being built into the operating model.
Regenemm has evolved into a hub-and-spoke architecture where domain-rich clinical workflows connect to a governed platform backbone.
Creative Thinking Institute has undertaken a multi-year research and development program for Regenemm, supported through the Australian Government's Department of Industry, Science and Resources R&D framework across 2020-2024.
This work focused on the technical challenge of combining biometric, psychometric, voice, clinical and contextual data into actionable healthcare insights. The program included experimentation in wearable data integration, real-time clinical audio processing, speaker diarisation, AI-assisted summarisation, hallucination reduction, multimodal synchronisation and clinical workflow design.
Regenemm Healthcare is being built with compliance and governance embedded into its operating model. CTI works with Vanta to support its formal accreditation and assurance pathway across ISO 27001, ISO 42001, HIPAA, GDPR, and SOC 2 readiness.
The operating principle is simple: no evidence means not done. Healthcare AI must be secure, auditable, explainable, and accountable from the beginning, not retrofitted after product-market fit.
CTI's role is to keep the parent-company narrative visible: why the system exists, what Regenemm is allowed to do, how transparency is expressed publicly, and where product-specific trust evidence belongs.
That means executive oversight, AI impact assessments, lifecycle gates, system cards, data lineage, release approvals, monitoring records, incident evidence, and Vanta-linked risk ownership.
Regenemm combines clinical AI assistants, structured documentation, patient-facing summaries, consent-aware workflows, interoperability, auditability, and compliance-led deployment.
AI is not a feature bolted onto the product. It is part of the clinical workflow architecture, data model, governance layer and operating system of the platform.
Regenemm Voice helps transform clinical conversations into structured documentation and patient-facing summaries designed to support clinician review, patient understanding, handover quality, and continuity of care.
Regenemm Healthcare is being developed with clinical research discipline, human ethics preparation and medical-device-style risk management.
The research program is designed to examine documentation efficiency, note completeness, clinician workflow, handover quality, patient communication, biosensor-assisted perioperative risk assessment and safe integration into Australian healthcare settings. AI assists with documentation and organisation of clinical information; healthcare professionals review and approve clinical content before use.
Participant information and consent materials are being prepared around voice recording, AI processing, optional wearable data, de-identified research use and future contact.
Regenemm is being positioned for perioperative communication workflows across surgeons, anaesthetists, nurses, perioperative physicians and documentation teams.
Risk management work is being developed with ISO 14971-style thinking across availability, data processing, cybersecurity, bias, drift, overreliance, training and biosensor limits.
The research program targets multi-specialty perioperative documentation, Australian healthcare validation, biosensor integration and audit-grade interoperability.
Current thinking on AI in healthcare, clinical workflow, governance, and patient communication.

Clinical state is inferred from multiple evidence streams. The task is to understand what those streams reveal about change.

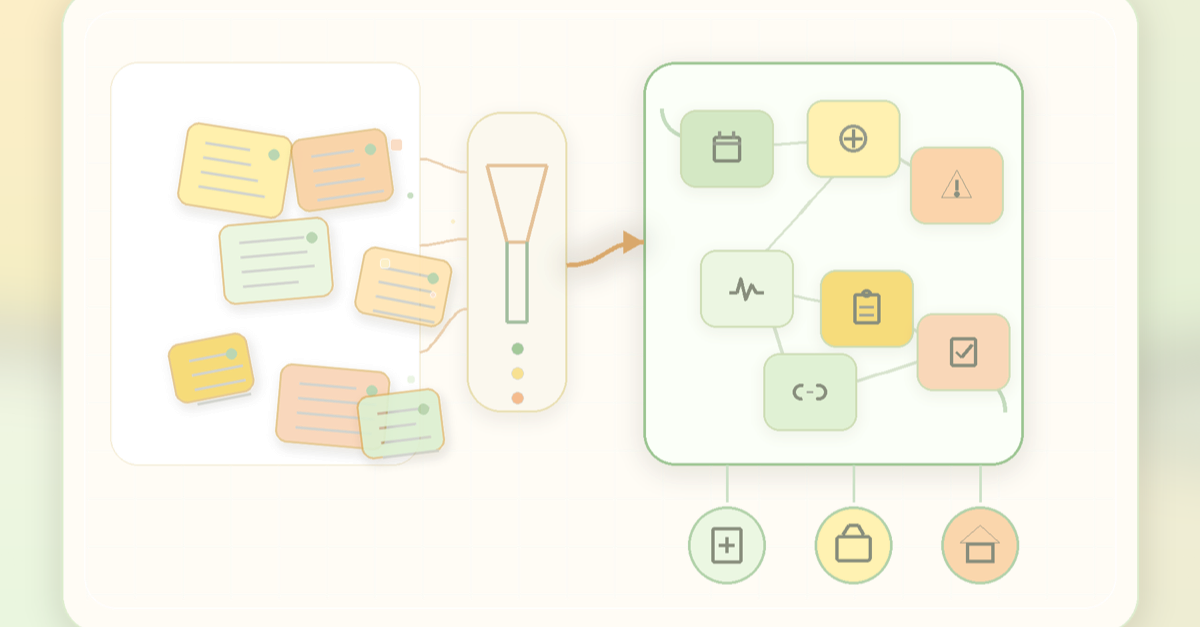
Narrative notes preserve nuance, but clinical intent needs structured FHIR pathways to travel across teams, systems and care settings.

Neurosurgical consultations are a demanding stress test for clinical AI because they combine imaging, examination, protected facts, irreversible decisions and medico-legal weight.

